Imagine walking into a pharmacy with a prescription for "acetaminophen." You hand it to the pharmacist, and they nod. Now, imagine you are in London, holding a script that says "paracetamol." To anyone who isn't a medical professional, these sound like two completely different chemicals. They aren't. They are the exact same medication. This confusion is exactly why generic naming conventions exist.
Drug names can be incredibly confusing. Every pill has at least two names: a brand name (like Tylenol) and a generic name (like acetaminophen). But even the generic names vary depending on where you live. In the United States, we use USAN. Globally, most of the world uses INN. Understanding the difference between these systems isn't just trivia for pharmacists; it’s critical for patient safety, avoiding dangerous medication errors, and understanding what you are actually putting into your body.
What Are USAN and INN?
To understand how drugs get their names, we have to look at the two main bodies responsible for creating them. These organizations work together, but they operate independently.
USAN stands for United States Adopted Names. It is the official nonproprietary naming system used in the United States. The USAN Council manages this process. This council is a unique partnership co-sponsored by three major organizations: the American Medical Association (AMA), the United States Pharmacopeial Convention (USP), and the American Pharmacists Association (APhA). When a new drug is developed for the US market, the manufacturer must apply to the USAN Council for an approved generic name.
INN stands for International Nonproprietary Name. This system is managed by the World Health Organization (WHO). Established in 1950, the INN program aims to create a single, universally recognized name for every pharmaceutical substance. If a drug has an INN, that name should theoretically be used everywhere in the world, from clinical trials to hospital prescriptions.
The goal of both systems is the same: to provide a unique, distinctive identifier for each drug substance. Unlike brand names, which are trademarks owned by companies, generic names belong to the public domain. No one can trademark "ibuprofen" or "lisinopril." This ensures that any company can manufacture and sell the drug under its generic name once patents expire.
How Do Drug Names Work? The Stem System
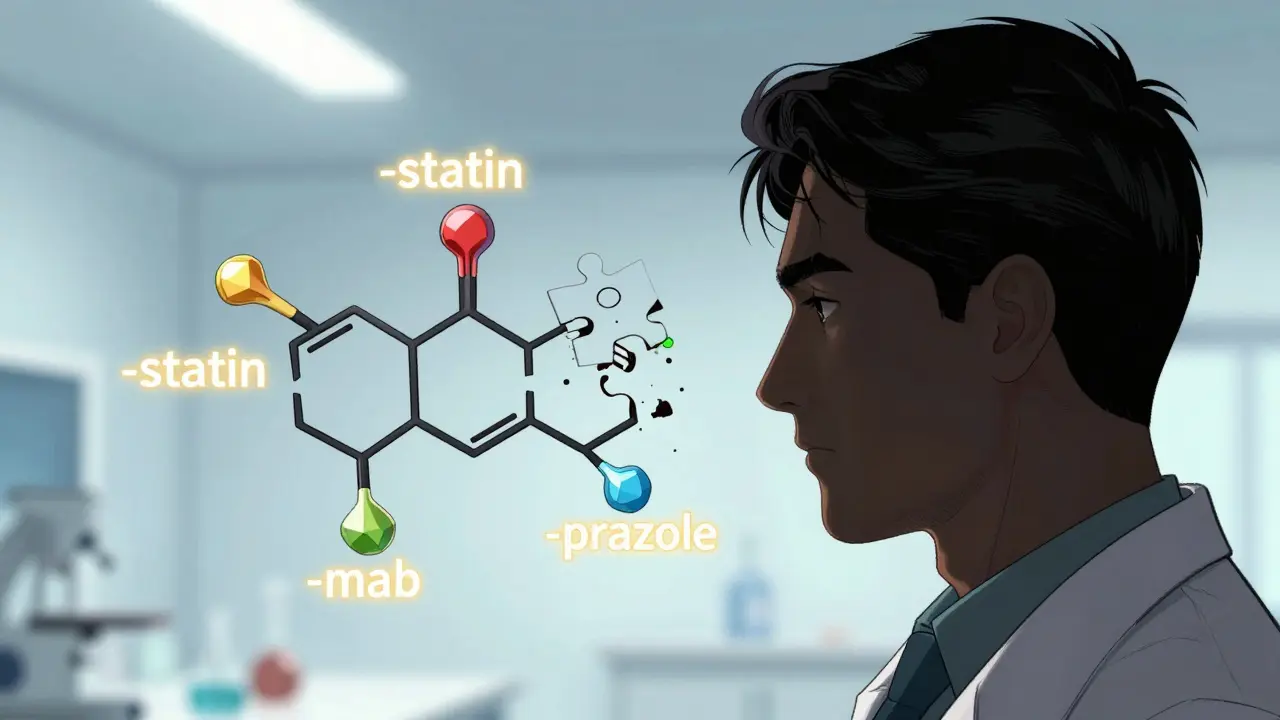
You might notice that many drug names end in similar ways. Drugs like atorvastatin, rosuvastatin, and simvastatin all treat cholesterol. They share the suffix "-statin." This isn't a coincidence. Both USAN and INN use a systematic "stem-based" naming convention.
The stem-usually the ending part of the word-tells you something about the drug's chemical structure, mechanism of action, or therapeutic class. Here is how it works:
- -prazole: Indicates proton pump inhibitors, used for acid reflux (e.g., omeprazole, pantoprazole).
- -statin: Identifies HMG-CoA reductase inhibitors, used to lower cholesterol (e.g., atorvastatin).
- -mab: Denotes monoclonal antibodies, often used in cancer or autoimmune treatments (e.g., trastuzumab).
- -virdine: Points to non-nucleoside reverse transcriptase inhibitors, used for HIV treatment (e.g., efavirenz).
- -feron: Relates to interferon compounds (e.g., peginterferon).
The prefix-the beginning part of the name-is usually a "fantasy" element. This is a string of letters chosen to sound pleasant and distinct, without carrying specific meaning. For example, in "omeprazole," "ome" is the fantasy prefix, and "prazole" is the stem. This combination creates a name that is unique but also gives healthcare providers a clue about what the drug does.
For complex biologics like monoclonal antibodies, the stems are even more detailed. According to WHO guidelines updated in 2021, the suffix indicates the type of antibody. For instance, "-ximab" refers to chimeric antibodies, while "-zumab" refers to humanized antibodies. This level of detail helps doctors quickly identify the nature of the therapy.
USAN vs. INN: Key Differences
While USAN and INN cooperate closely, they are not identical. In fact, there are notable differences that can cause confusion, especially for patients traveling or accessing international medical records.
| Feature | USAN (United States) | INN (Global) |
|---|---|---|
| Governing Body | USAN Council (AMA, USP, APhA) | World Health Organization (WHO) |
| Primary Region | United States | International (most countries) |
| Alignment Rate | ~95% aligned with INN | ~95% aligned with USAN |
| Exceptions | Acetaminophen, Albuterol, Rifampin | Paracetamol, Salbutamol, Rifampicin |
| Regulatory Requirement | Required by FDA for US marketing | Required by EMA and most global regulators |
About 95% of the time, the USAN and INN are the same. However, those remaining 5% represent significant historical exceptions. The most famous examples include:
- Acetaminophen (USAN) vs. Paracetamol (INN): The pain reliever known as Tylenol in the US is called paracetamol in Europe, Asia, and most of the rest of the world.
- Albuterol (USAN) vs. Salbutamol (INN): This common asthma inhaler has different names depending on whether you are in North America or elsewhere.
- Rifampin (USAN) vs. Rifampicin (INN): An antibiotic used to treat tuberculosis and other infections.
These differences exist because USAN sometimes retains names that were already established in US medical practice before the global standardization efforts intensified. The USAN Council states that it does not automatically adopt INN names if a different name is already entrenched in US usage. This can lead to medication errors in multinational healthcare settings, where a doctor might prescribe "salbutamol" and a US pharmacist might not immediately recognize it as albuterol.
The Naming Process: From Lab to Label
Getting a drug name approved is a rigorous, multi-step process that takes time. Pharmaceutical companies cannot simply pick a name and start selling the drug. They must navigate a strict approval timeline.
- Application Submission: Manufacturers typically submit naming applications during Phase 1 or early Phase 2 clinical trials. This timing is crucial because the approval process takes approximately 18 to 24 months.
- Proposal of Names: The company submits up to six proposed names in order of preference. These names must be free of trademark conflicts and distinct from existing generic or brand names.
- Review and Verification: USAN Program staff verify the chemistry of the drug and search databases for potential conflicts. They prepare a ballot with the firm's proposals and any alternative suggestions generated by the council.
- Voting and Acceptance: The USAN Council votes on the names. Once a name is accepted, it is submitted to the WHO's INN Programme for international review.
- Public Objection Period: Accepted names are published for a mandatory four-month objection period. During this time, any stakeholder can raise concerns about the name's distinctiveness or potential for confusion.
- Final Adoption: If no objections are raised-which is rare due to the thorough prior screening-the name becomes officially recognized.
This process is expensive and time-consuming. Industry experts note that companies often go through 15 to 20 name iterations before finding a viable candidate. Many names are created and discarded to ensure the final choice is distinctive enough to prevent medication errors.
Why Distinctive Names Matter for Patient Safety
The primary goal of pharmaceutical nomenclature is patient safety. Medication errors related to name confusion cost the US healthcare system approximately $2.4 billion annually. A significant portion of these errors stems from look-alike or sound-alike drug names.
Consider the difference between a brand name and a generic name. Brand names are designed to be memorable and marketable. They are often short, catchy, and easy to pronounce. However, this can backfire. If two brand names sound too similar (e.g., Xanax and Ativan), doctors might misread a handwritten prescription, or patients might confuse them verbally.
Generic names, by contrast, are designed to be distinctive and informative. The stem system helps here. If a doctor sees a name ending in "-mab," they know it is a monoclonal antibody. If they see "-pril," they know it is an ACE inhibitor for blood pressure. This immediate recognition reduces the risk of prescribing the wrong class of drug.
However, the system is not perfect. As drug mechanisms become more complex, traditional stems may not adequately capture nuanced therapeutic actions. For example, some newer drugs have multiple mechanisms of action that don't fit neatly into existing categories. The USAN Council acknowledges this challenge, noting that stems are defined based on available knowledge at the time of naming. Even if a drug's indication evolves later, the name usually remains unchanged.
Challenges with New Therapies
The pharmaceutical industry is moving rapidly toward biologics, gene therapies, and RNA-based therapeutics. These new modalities pose significant challenges to traditional naming conventions.
Biologics now represent 42% of global pharmaceutical sales, amounting to approximately $380 billion in 2023. Unlike small-molecule drugs (like aspirin), biologics are large, complex proteins produced in living cells. Their mechanisms are often less straightforward than simple enzyme inhibition.
The WHO updated its monoclonal antibody naming conventions in 2021 to accommodate newer formats. But emerging therapies like antibody-drug conjugates (ADCs) and gene editors don't fit easily into existing stem categories. The USAN Council and WHO are currently developing frameworks to handle these complexities. They aim to create new stems only when existing schemes fail to accurately represent a compound's action or structure, and only when substantial preclinical data support the change.
This evolution is critical. As more personalized and targeted therapies enter the market, the need for clear, unambiguous naming becomes even more vital. A confusing name for a gene therapy could have dire consequences, given the high stakes and specialized nature of these treatments.
Practical Tips for Patients and Providers
Navigating drug names can be tricky. Here are some practical tips to help you stay safe and informed.
- Know Both Names: Always ask your pharmacist for both the brand name and the generic name of your medication. This helps you recognize the drug if you travel or switch insurance plans.
- Check for Stems: If you take multiple medications, check if they share the same stem. Taking two drugs with the same stem (e.g., two "-statins") might indicate duplication or interaction risks. Consult your doctor.
- Be Aware of Regional Differences: If you travel internationally, bring a list of your medications with both USAN and INN names. This prevents confusion at foreign pharmacies.
- Verify Prescriptions: If a prescription looks unclear or sounds like another drug you take, call your pharmacist immediately. Don't assume it's correct.
- Use Reliable Sources: Refer to official resources like the FDA label or the WHO INN database for accurate information. Avoid relying solely on internet searches, which may contain outdated or incorrect data.
What is the difference between USAN and INN?
USAN (United States Adopted Names) is the official generic naming system used in the United States, managed by the USAN Council. INN (International Nonproprietary Name) is the global standard managed by the World Health Organization (WHO). While they align about 95% of the time, some drugs have different names in the US versus the rest of the world, such as acetaminophen (USAN) vs. paracetamol (INN).
Why do drugs have stems in their names?
Stems are suffixes added to drug names to indicate their therapeutic class, mechanism of action, or chemical structure. For example, "-statin" indicates a cholesterol-lowering drug, and "-prazole" indicates a proton pump inhibitor. This helps healthcare professionals quickly identify the type of drug and reduces the risk of medication errors.
Can a company trademark a generic drug name?
No. Generic names, whether USAN or INN, are in the public domain. No individual or company can own or trademark them. This ensures that any manufacturer can produce and sell the drug under its generic name once patents expire, promoting competition and lower prices.
How long does it take to get a drug name approved?
The naming process typically takes 18 to 24 months. Companies usually submit applications during Phase 1 or early Phase 2 clinical trials to ensure the name is ready by the time the drug reaches the market. The process involves multiple reviews, database checks for conflicts, and a public objection period.
What are some common examples of USAN and INN differences?
Common examples include acetaminophen (USAN) vs. paracetamol (INN), albuterol (USAN) vs. salbutamol (INN), and rifampin (USAN) vs. rifampicin (INN). These differences reflect historical usage patterns and regional medical practices.
Who manages the USAN naming system?
The USAN Council manages the system. It is co-sponsored by the American Medical Association (AMA), the United States Pharmacopeial Convention (USP), and the American Pharmacists Association (APhA). The council ensures that names are distinctive, scientifically accurate, and free of conflicts.
Why is patient safety a priority in drug naming?
Medication errors due to name confusion cost the US healthcare system billions annually. Distinctive names help prevent misreading prescriptions, mixing up similar-sounding drugs, and administering the wrong medication. The stem system aids in quick identification of drug classes, further enhancing safety.

